Official Journals By StatPerson Publication
|
Table of Content - Volume 12 Issue 2 - November 2019
Establishment of reference range for maternal thyroid function of Gujarati women
Arpita Patel1, Margit Gajjar2*, Dipti Gajjar3
1Department of Biochemistry, Medical College Baroda, Raopura, Vadodara 390001, Gujarat, INDIA. 2Department of Biochemistry, 3Department of Pathology, GMERS Medical College, Kharvad ground, Sipor road, Vadnagar 384355 Gujarat, INDIA Email: drappupatel@gmail.com,margitgajjar@gmail.com, diptikrs@gmil.com
Abstract Objective: Pregnancy is a state of stress for the thyroid, resulting in hypothyroidism in women with limited thyroidal reserve or iodine deficiency and increases fetal demand. Therefore, nonpregnant reference intervals should not be used in pregnancy as they can mislead on the diagnosis and treatment of thyroid disorders during pregnancy. The aim of this study was to establish reference range for thyroid hormone during pregnancy in a tertiary care center in Gujarat. Design and Methods: A total of 432 pregnant women and 262 nonpregnant women were tested for thyroid profile which included TSH, TOTAL T4 and TOTAL T3. Estimation for T3, T4 and TSH were done using the ELISA. Result: The study population comprised 694 women, 432(68%) were pregnant and 262(32%) were non-pregnant. The RIs of T3, as obtained in our study, are 1.5 to 2.0 ng/mL for the pregnant women; 1.0 to 1.4 ng/mL for the non-pregnant women Similarly, the RIs of T4 for pregnant and non-pregnant women were 7.1 to 10.6 and 6.1 to 10.1 μg/dL respectively, and the RIs of TSH for pregnant and non-pregnant women were 0.6 to 3.8 and 1.3 to 4.1 mIU/L respectively. Conclusion: Study provides reference intervals of thyroid hormones in pregnant Gujarati women. It is important that each laboratory in our country should determine its own population and method-based Reference Interval (RI) for diagnosing gestational thyroid disorders. Key words: Pregnancy, TSH, T3, T4, Reference Interval
Pregnancy has a profound impact on the thyroid gland and its function. During pregnancy, the thyroid gland increases in size by 10% in iodine replete countries but by 20% to 40% in areas of iodine deficiency. Production of the thyroid hormones, thyroxine (T4), and triiodothyronine (T3), increases by nearly 50%, in conjunction with a separate 50% increase in the daily iodine requirement. These physiological changes happen seamlessly in healthy women, but thyroid dysfunction can occur in many pregnant women because of pathologic processes.1 Thyroid dysfunction during pregnancy is common, with a prevalence of 2%–4%.2,3 Maternal thyroid dysfunction is associated with an increased risk of various adverse maternal and child outcomes, including miscarriage, intrauterine growth retardation, hypertensive disorders, preterm delivery, and a decreased child IQ 4–5. Pregnancy is associated with a number of physiological and hormonal changes that result in significant but reversible alterations in thyroid function tests (TFTs). Increasing attention has, therefore, focused on the diagnosis and treatment of maternal thyroid dysfunction during pregnancy. Therefore, nonpregnant reference intervals should not be used in pregnancy as they can mislead on the diagnosis and treatment of thyroid disorders during pregnancy. Establishment of reference intervals in pregnancy is thus of great importance, and these values should be used to diagnose thyroid disorders during pregnancy. However, many factors such as ethnicity, age, manufacturer's methodology, iodine status, and rigor for selection of the reference population and calculation method may affect the establishment of reference intervals for TFTs. Since reference intervals needs to be gestational age, method, and population specific, there is need to establish thyroid levels in pregnancy for different population across the world. In India, limited data's are available over thyroid hormones level during pregnancy and none from Gujarat.6,7,8,9 The aim of this study was to establish reference range for thyroid hormone during pregnancy in a tertiary care center in Gujarat.
MATERIAL AND METHOD This was a study conducted at the Department of Biochemistry, S.S.G. Hospital, Vadodara, Gujarat. A total of 432 pregnant women and 262 nonpregnant women were tested for thyroid profile which included TSH, TOTAL T4 and TOTAL T3. As this is retrospective analysis of the data, approval of ethical committee doesn’t require. Data, used to determine reference interval extracted from the laboratory information system (LIS) running at the Clinical Biochemistry laboratory, S.S.G. Hospital, Vadodara, Gujarat. Exclusion criteria Women having presence of anti-thyroid peroxidase antibody (TPO antibodies) were excluded. Estimation for T3, T4 and TSH, Anti-thyroid peroxidase antibody (TPO antibodies) were done using the ELISA technique using commercially available kits by ERBA, MENHEIM on ELISA MACHINE. Statistical Analysis All data were tabulated into MS Excel and analyzed using Medcal C software version 19.0.4. Distribution nature of T3, T4, and TSH values of pregnant and non-pregnant women were observed by inspecting histograms. For pregnant and non-pregnant women, mean and standard deviation of T3, T4, and TSH were calculated separately. The RI (2.5-97.5th percentile) was estimated using nonparametric estimation combined with bootstrap resampling.
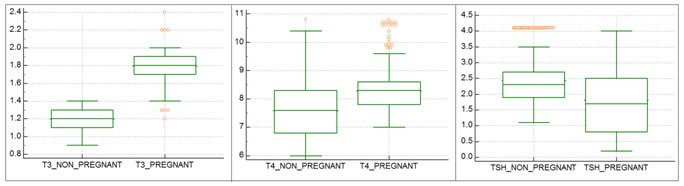
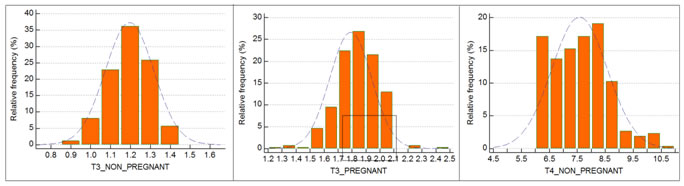
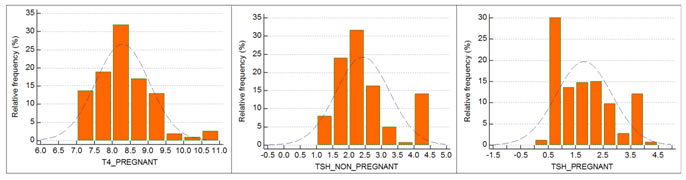
RESULT The study population comprised 694 women, 432 (68%) were pregnant and 262(32%) were non-pregnant. After execution of the exclusion criteria, 9 women were excluded from the study. The age of the women ranged from 18 to 40 years. We have calculated mean, standard deviation for TSH, T3 and T4. Which are tabulated in table no 1.A reference interval for each parameter was calculated to obtain 2.5th and 97.5th percentile. The RIs of T3, as obtained in our study, are 1.5 to 2.0 ng/mL for the pregnant women; 1.0 to 1.4ng/mL for the non-pregnant women (Table 3). Similarly, the RIs of T4 for pregnant and non-pregnant women were 7.1 to 10.6 and 6.1 to 10.1 μg/dL respectively, and the RIs of TSH for pregnant and non-pregnant women were 0.6 to 3.8, and 1.3 to 4.1 mIU/L respectively (Table 3). Box plots of T3, T4 and TSH values for pregnant and non-pregnant are depicted in Graphs 1 to 3 respectively. Frequency distribution of T3, T4 and TSH of pregnant and nonpregnant women also showed in subsequent graphs. The RI values provided by kit manufacturer were generalized and not specific for pregnancy/trimester wise. The RIs of T3, T4, and TSH as supplied by the kit manufacturer were 0.51-1.58 ng/mL, 5.3 to 12.1 μg/dL, and 0.5-5.5 mIU/L. (Table 2).
Graph 1 Graph 2 Graph 3 Graph 1: Box plots for T3 values of Pregnant and Non-pregnant women; Graph 2: Box plots for T4 values of Pregnant and Non-pregnant women; Graph 3: Box plots for TSH values of Pregnant and Non-pregnant women.
Table 1: Mean± 2SD of TSH, T3 and T4 in Pregnant and Non-pregnant women
Table 2: Descriptive statistics of reference population segregated into Pregnant and Non-pregnant women Table 3: Reference data for T3, T4, and TSH according to our study
a bootstrap confidence interval ( 10000 iterations; random number seed: 978)
Table 4: Reference intervals for TSH, T3 and T4 by different studies
DISCUSSION It has been shown that the physiological condition like pregnancy can alter the thyroid hormone levels and thyroid hormone reference range for children and pregnant mothers is different. Hence, selection and proper interpretation of gestational thyroid function parameters are important for the wellbeing of both the mother and the fetus. 9 Various researches have been conducted to find out the prevalence of thyroid disorder in different states of India, but it appears that there have been very few studies carried out in Vadodara, Gujarat area. 10, 11 Pregnancy is a crucial stage in the life of a woman. Untreated and undetected gestational thyroid disorders are known to cause many complications like anaemia, pregnancy-induced hypertension, haemorrhage, preterm delivery in mothers and low birth weight, prematurity, congenital malformation, intrauterine fetal death, and lower intelligence quotient in children. 12 B.Chakrabarty et. al. derived RIs for T3, T4 and TSH in pregnant women were 1.21-1.51 ng/ml, 7.57-8.44 μg/dL and 2.26-2.85 mIU/L respectively 13 Table 4 shows reference intervals for T3, T4 and TSH in different studies. On comparing these RIs obtained and the kit manufacturer RIs, an appreciable variation of interpretation of test results was seen. This variation is because the RI provided by the reagent manufacturer has been determined on age, sex nonspecific, and nonpregnant population and these reference intervals cannot be applied for the interpretation of thyroid function tests of a pregnant woman. According to the IFCC guidelines of reference values and recommendations of the Indian Thyroid Society in the line of American Thyroid Association, RI in healthy population is to be determined using stringent specific exclusion criteria. 1, 14 In order to fit this criterion, we have used stringent criteria for our reference population selection. Our assessments were done by the most commonly used ELISA method with standard instrument and standard kits (ERBA MENNHEIM, GERMANY). Tests were done with proper quality control measures. The statistical strengths are determined by distributions examination and application of proper parametric or nonparametric methods.
STRENGTH The strength of our study is that it is based on the population migrated to Vadodara for professional purpose and resulting in representation of ideal pan Indian reference population which is having pan Indian profile because of multicultural and multiethnicity and demographic distribution. We have assessed TSH, total T3 and total T4 levels by ELISA method, which is most commonly assessed, economical, and most readily available parameter in our country.
LIMITATIONS There are some limitations to this study. Maternal TSH, T3 and T4 values in each trimester were not measured serially in the same pregnant women. Further follow-up study with more stringent inclusion and exclusion criteria will improve our strength of data to provide more conclusive trimester-specific levels and FT4 measurement would be more accurate than Total T4. Trimester specific reference value can be studied using larger sample size. Second, we did not study the long-term neuropsychological development of children born from mothers with different levels of serum TSH, T3 and T4.
CONCLUSION This study provides reference intervals of thyroid hormones in pregnant Gujarati women that differed from those of pregnant women in other regions of the India and different countries with different ethnicity and iodine nutrition. It is important that each laboratory in our country should determine its own population and method-based RI for diagnosing gestational thyroid disorders. To promote this concept, we have determined our own laboratory specific, method-specific; RI for the most commonly used thyroid hormone parameters level by ELISA method and intends to use them for screening of Indian pregnant mothers. In addition, education about adequate iodine intake during pregnancy to prevent both inadequate and excessive iodine intake must be emphasized.
REFERENCE
|
 Home
Home